Gas compression is a process that increases the pressure of a gas by reducing its volume, typically achieved through the use of compressors. This is commonly employed in various industrial applications, such as in natural gas processing, refrigeration, and power generation. In this article, we will delve into the fundamentals of gas compression, its practical applications in engineering, and the types of gas compressors.
Fundamentals of Gas Compression
Gas compression is the process of reducing the volume of a gas while increasing its pressure. This process is essential in various engineering applications such as pumps, compressors, and turbines.

Gas compression can occur under different thermodynamic conditions: isothermal, adiabatic, and polytropic.
In an isothermal compression process, the temperature of the gas remains constant throughout the compression. Achieving isothermal compression typically involves a slow compression process, allowing for heat exchange with the surroundings to maintain a constant temperature. The relationship between pressure and volume during isothermal compression is described by Boyle’s Law as follows:

Elevate Your Engineering With Excel
Advance in Excel with engineering-focused training that equips you with the skills to streamline projects and accelerate your career.
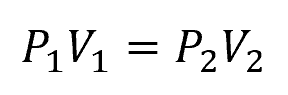
Where:
- P1, P2 = initial and final pressures [Pa]
- V1, V2 = initial and final volumes [m3]
On the other hand, adiabatic compression involves a rapid process where there is no heat exchange with the surroundings. The temperature of the gas changes without heat entering or leaving the system. The relationship between pressure and volume in this case is described by the adiabatic equation:
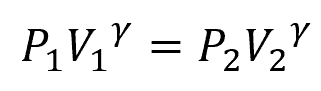
Where:
- γ = adiabatic index [unitless]
The adiabatic index is the ratio of specific heats:
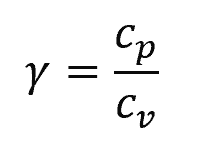
Where:
- cp = specific heat capacity at constant pressure [J/kg-K]
- cv = specific heat capacity at constant volume [J/kg-K]
However, achieving true adiabatic conditions is only possible under ideal research conditions. In practice, heat is rejected or added in compressors due to leakage and friction. In commercial compressors, the process is neither adiabatic nor isothermal, but falls somewhere between the two, known as a polytropic process.
A polytropic compression process is more general and is characterized by the equation:
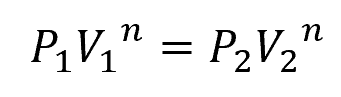
Where:
- n = polytropic index [unitless]
The value of the polytropic index depends on the design of the compressor and is determined from actual performance data.
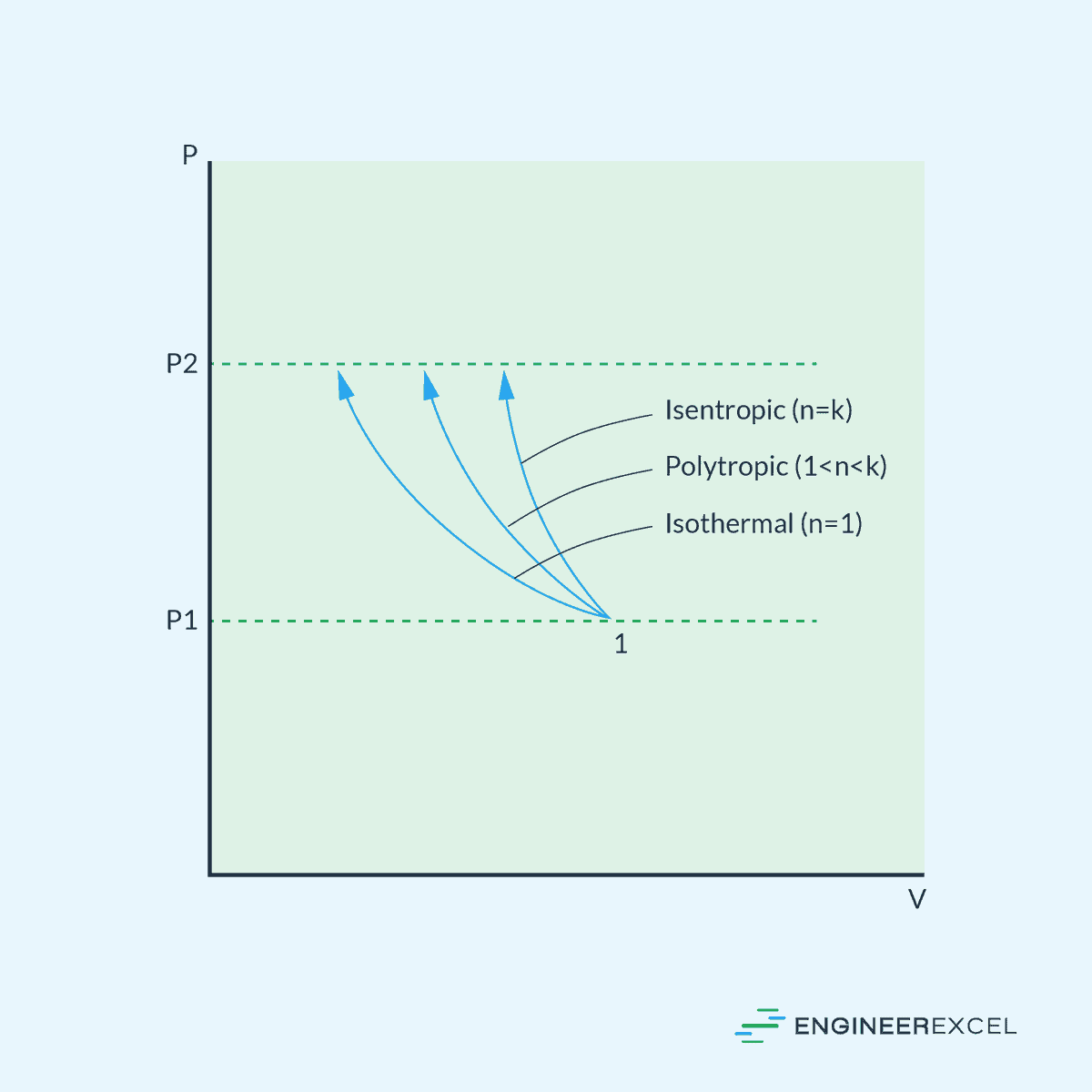
The diagram below compares the pressure-volume behavior of the different types of gas compression processes.
Applications of Gas Compression
Natural Gas Transportation
In the natural gas industry, gas compression serves the purpose of increasing the pressure of natural gas for efficient transportation through pipelines. Compressed gas is easier to transport with reduced volume and lower energy loss. The compression process involves a series of reciprocating compressors strategically placed along the pipeline to maintain the desired gas pressure.
Refrigeration and Air Conditioning
Refrigeration and air conditioning systems rely on the principle of gas compression for their operation.
The refrigerant gas is compressed in a compressor, raising its temperature and pressure. The hot compressed gas then condenses in a heat exchanger, releasing thermal energy to the surroundings. The condensed liquid refrigerant expands through an expansion valve, reducing its pressure and temperature, resulting in the evaporation and cooling of the refrigerant in the evaporator.
Industrial Processes
Gas compression is also utilized in many industrial processes that require high-pressure gases in their operations. Examples of such processes include:
- Chemical synthesis: Certain chemical reactions require high pressure and temperature to occur at a reasonable rate. Gas compression provides the necessary conditions for these reactions to take place.
- Power generation: Gas turbines and reciprocating engines often use compressed air or gas mixtures as input to generate electricity.
- Pneumatic tools and equipment: Compressed air is used in various tools and equipment, such as air hammers, spray guns, and pneumatic actuators.
Types of Gas Compressors
There are two main types of gas compressors: dynamic compressors and positive displacement compressors. Each type has its own working principle, advantages, and disadvantages.
Dynamic Compressors
Continuous flow or dynamic compressors increase the gas pressure by imparting kinetic energy to the gas particles and converting it into static pressure. There are two primary types of dynamic compressors:
- Centrifugal Compressors: These compressors use a rotating impeller to accelerate the gas and then decelerate it in a diffuser, which converts the kinetic energy into pressure. They are best suited for continuous flow applications and can accommodate large volumes of gas. Centrifugal Compressor Applications include refrigeration, pipeline compression, and gas lift.
- Axial Flow Compressors: Gas flows parallel to the rotor axis, passing through several stages of rotating and stationary airfoils. These compressors work well in high flow rate and medium pressure applications, such as jet engines and industrial gas turbines.
Positive Displacement Compressors
Positive displacement compressors force a specific volume of gas through a confined space to mechanically increase the pressure. These compressors are further classified into two main types:
- Reciprocating Compressors: These compressors use a piston inside a cylinder to compress the gas, similar to an automobile engine. Reciprocating compressors typically have higher pressure ratios and are suitable for intermittent flow applications. They are most commonly found in petroleum refining, natural gas processing, and refrigeration systems.
- Rotary Compressors: Rotary compressors consist of rotating elements (e.g., screws, vanes, or lobes) to compress the gas. These compressors provide continuous, smooth flow without any significant pulsation. They are widely used in HVAC applications, industrial compressed air systems, and gas processing plants.
